ss-31
the mitochondrial repair expert
overview
SS-31 (Elamipretide)
1. What is SS-31?
SS-31 (commonly known in clinical literature as Elamipretide, Bendavia, or MTP-131) is a synthetic, cell-penetrating tetrapeptide (a sequence of four amino acids: D-Arg-Dmt-Lys-Phe-NH2).
Unlike most peptides that interact with cell surface receptors, SS-31 is specifically engineered to be mitochondria-targeted. Because of its unique alternating cationic (positively charged) and aromatic structural properties, it easily crosses the cell membrane and selectively accumulates in the inner mitochondrial membrane (IMM).
2. Mechanism of Action: Re-energizing the Cell
To understand SS-31, you have to understand Cardiolipin.
Cardiolipin is a unique lipid found exclusively in the inner membrane of mitochondria. It acts like the structural "glue" that holds the Electron Transport Chain (ETC) complexes together. When the ETC is tightly packed, the mitochondria produce ATP (energy) efficiently.
When cells experience aging, stress, or disease, excess Reactive Oxygen Species (ROS) damage the cardiolipin. The ETC complexes drift apart, ATP production crashes, and a vicious cycle of oxidative stress begins.
How SS-31 Fixes This:
Selective Binding: SS-31 selectively binds directly to cardiolipin.
Structural Stabilization: By binding to cardiolipin, SS-31 restores the physical shape of the inner mitochondrial membrane.
Efficiency Restoration: It pushes the Electron Transport Chain complexes back together, restoring healthy electron transfer.
ROS Reduction: Because the electron transport chain is no longer "leaking" electrons, the production of toxic reactive oxygen species drops dramatically.
In short: SS-31 doesn't just act as a traditional antioxidant that cleans up waste; it fixes the engine so the waste isn't produced in the first place.
3. Key Research Applications and Benefits
Based on pre-clinical and clinical trial data, SS-31 is heavily researched for its neuroprotective and anti-aging properties:
Mitochondrial Myopathy & Sarcopenia: Reverses age-related muscle wasting and improves exercise tolerance.
Neuroprotection: Demonstrates ability to cross the blood-brain barrier to reduce neuroinflammation, improve memory impairment, and protect against traumatic brain injury (TBI).
Cardiovascular & Renal Health: Protects tissues from ischemia-reperfusion injury (tissue damage caused when blood supply returns to tissue after a period of lack of oxygen).
Macular Degeneration: Currently researched for reversing ocular decline associated with mitochondrial dysfunction.
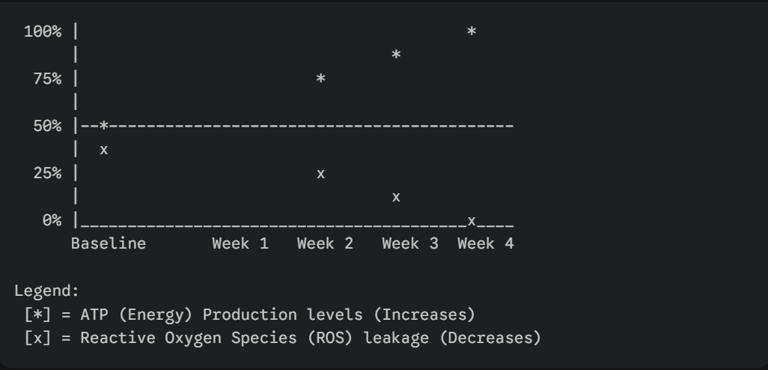
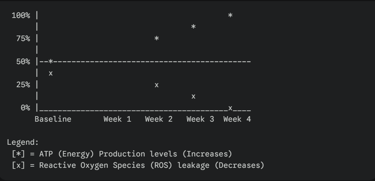
Graph: Mitochondrial ATP Production vs. Oxidative Stress (Over Time with SS-31) This graph illustrates the inverse relationship between energy production and cellular stress once SS-31 is introduced.
Disclaimer: This information is for educational purposes only.